copyright © ONERA 1996-2006 - All rights reserved
Microstructure of a niobium silicide based alloy, with Nb5Si3 dendrites (dark phase) surrounded by a mixture of niobium (light phase) and Nb5Si3.
Image: J.L. Raviart, S. Drawin, DMSM
* : refer to Glossary
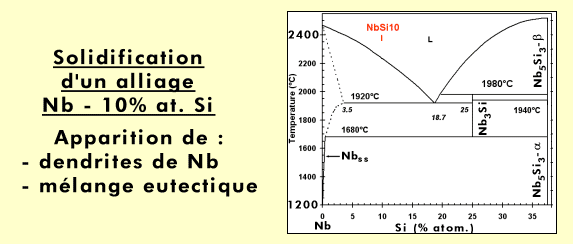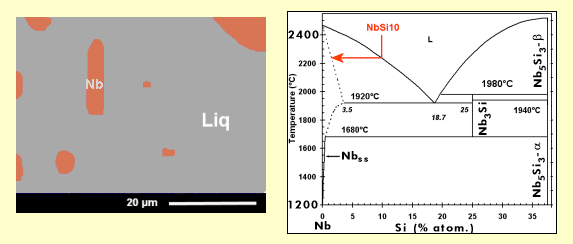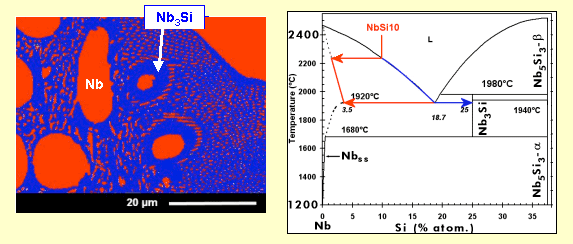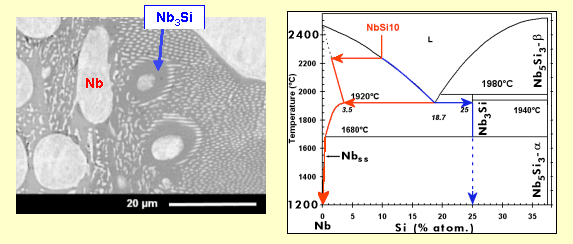
The material has been obtained by arc-melting* a mixture of pure elements (Nb, Si and refractory* metals). This processing induces a relatively rapid cooling, with the consequence that, in a binary Nb-Si alloy, the metastable* Nb3Si is retained at ambient temperature, although a Nb + Nb5Si3 is expected according to the phase diagram below.
This Nb3Si phase (which is not desired because of bad mechanical properties) can be transformed into Nb5Si3 (through the reaction 3 Nb3Si --> 4 Nb + Nb5Si3) if a subsequent heat treatment is applied. But the addition of some metallic elements to the starting materials can also directly force the formation of Nb5Si3 during solidification.
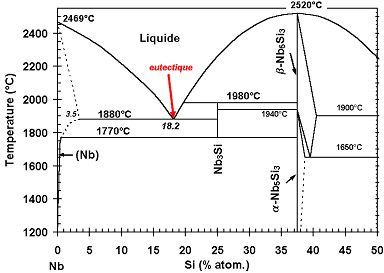
Nb-Si partial phase diagram (0 =< Si (% at .) =< 50).
During cool-down of a melt (see animation, in French), small crystals (solid nuclei) are formed, which grow continuously to finally yield dendrites*. When the melt, with a composition corresponding to the eutectic* (18.2 atom-% of silicon), reaches the “eutectic temperature” (here 1880°C), all the liquid solidifies instantaneously into a mixture of niobium (containing a small amount of silicon) and Nb3Si, with a specific microstructure, known as “eutectic”: one phase (here Nb) is finely dispersed in form of rods (it is the case here; the rod diameter is less than 500 nm) or platelets in the other phase. Depending on the angle at which the rods have been cut (for observation in the scanning electron microscope), they appear as circular or more or less elongated elliptical shapes.

Eutectic microstructure of a Nb (81.8%) – Si (18.2%) (atom percent) alloy: niobium (light phase) dispersed in Nb3Si (grey phase).
Photograph: A. Bachelier, DMSM.

High magnification view of the eutectic microstructure in a Nb (81.8%) – Si (18.2%) alloy (Nb is the light phase, Nb3Si is the dark phase).
More...
Refractory metal silicide based materials are developed to allow manufacture of aircraft or rotorcraft turbine components (e.g. blades and vanes) that could withstand very high temperature, i.e. about 1300°C compared to the ca. 1100°C for the currently used nickel based single crystal superalloys*. The increased temperature capability will allow reduction of specific fuel consumption, CO2 emissions and cooling air requirement, which will lead to further increases in efficiency and reductions in component weight.
At ONERA, part of the activities in this field is performed in the frame of the Ultmat project (“Ultra high temperature material for turbines”) which is funded by the European Commission. Started in January 2004 for a period of four years, this project will evaluate the potential of molybdenum and niobium silicide based alloys. The work programme includes definition of alloy compositions, extensive characterisation, and development of industrial scale processing, manufacturing and machining techniques to propose a new generation of materials exhibiting an acceptable trade-off between mechanical properties (at low and high temperature) and oxidation resistance.
Author:
Stefan Drawin is senior research engineer in the Metallic Materials and Processing Department [DMSM]. He is coordinator of the European Ultmat project.
Glossary
- Arc-melting
Processing technique where an electric arc is produced between the material to be melted (which has to be an electrical conductor) and a tungsten electrode. - Dendrite
Crystal , usually obtained by solidification, with a tree shape. A snow flake, for instance, has a dendritic structure. - Eutectic [from the Greek eutektos, "which melts easily"]
A eutectic is a mixture of several compounds (here Nb and Nb3Si) at a composition where melting and solidification occurs at a given temperature, unlike usual mixtures (for which each compound melts at its own “melting temperature”). This temperature is lower than that of any other mixture of the same compounds. It is an invariant point in a phase diagram (i.e. temperature and composition are fixed). - Metastable
Property of a phase that is observed although it not thermodynamically stable. Its evolution towards a stable state is slowed down (or even stopped) by very sluggish kinetics. - Refractory
Characterises a material that withstands high temperatures. - Superalloy
Alloy with about ten metallic elements, exhibiting at high temperature far better mechanical properties and oxidation resistance than the best steels.